Lack of Adherence Data – Is Pharma in the Dark?
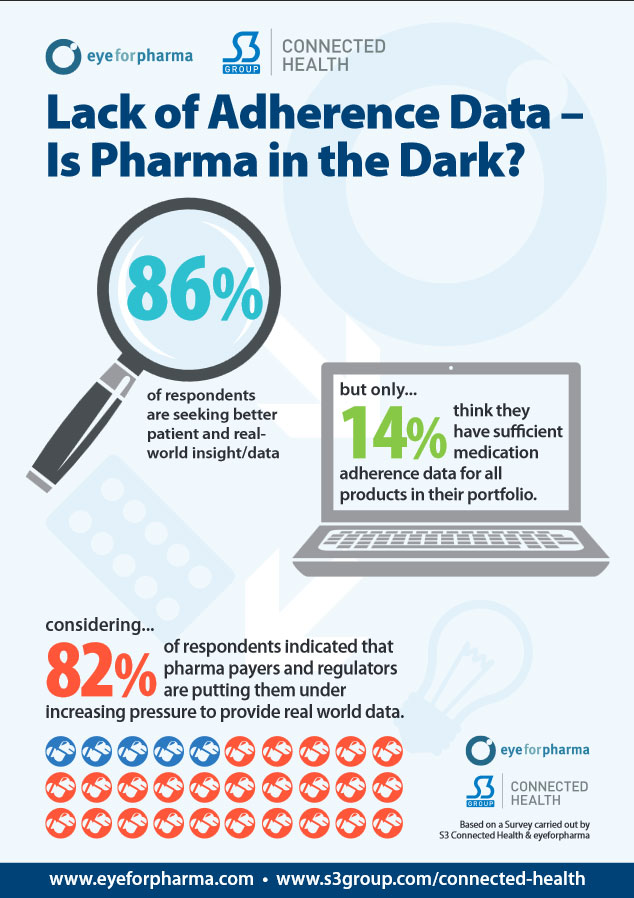
A mere 14% of pharma think they have sufficient medication adherence data for all products in their portfolio says S3 Survey.

We live in a world where consumer and customer insight is hugely valuable – where data is the raw material for companies like Google and Facebook. Indeed, organizations across sectors like airlines, retail and insurance are driven to a large extent by data they have on customer and market behavior. Compare this with the relative lack of data on the commercial side of the pharma industry. If data and customer insight casts light on your business, then it would appear that pharma are still using candles.
eyeforpharma and S3 Group, specialists in applying connected technology to promote adherence and support patients, recently conducted a survey on the topic of pharma’s attitude to the issue of medication non-adherence. The survey found that pharma are getting serious about the problem - but they are also reporting a lack of data to help them solve it.
Our survey showed that 86% of respondents are motivated to consider medication adherence solutions and are seeking better patient and real-world insight/data. Yet, only 14% of respondents think they have sufficient medication adherence data for all products in their portfolio.
Please click here for the full infographic.
This data is elusive because of the regulatory constraints on pharma, which make it difficult for them to interact with patients and gather data outside of a clinical trial setting. This real world data is becoming more important for pharma companies, with 82% of respondents indicating that pharma payers and regulators are putting them under increasing pressure to provide real world data.
The shift towards value pricing models in the market also increases the need for evidence of outcomes. In our survey, 3 out of every 4 respondents pointed to the increasing importance of these models.
More progressive organizations are putting down foundations which will enable them to offer a variety of compelling services and generate the real world data required to satisfy payers.”
Jim O’Donoghue, Head of Connected Health in S3, says, “Payers are pushing back on pharmaceutical companies and demanding more evidence of differentiated outcomes and value for money – they are increasingly moving toward a pay-for-performance model. Currently, pharma companies are not well equipped to provide the type of evidence required. We are working with a number of organizations moving toward the provision of data-rich patient services. Some are dipping their toe in the water but more progressive organizations are putting down foundations which will enable them to offer a variety of compelling services and generate the real world data required to satisfy payers.”
While the need to support market access and reimbursement negotiations is the primary commercial driver for initiating adherence programs, the data generated can also be used as input back into the product development process - helping pharma companies identify new opportunities for product differentiation and innovation.
“Data is an increasingly important resource throughout the lifecycle of a drug – the ability to create and leverage it to deliver better patient and commercial outcomes is as fundamental to pharma success as the microscope,” says O’Donoghue.
Register here to receive the full report of the survey as soon as it becomes available. The report provides some interesting insights into how pharma are tackling the very real and costly problem of adherence.