Market Access & Pricing Excellence 2015
Co-create solutions with payers. Get reimbursed for real patient value.
The Price of Global Health: A guide to access and pricing
Ed Schoonveld, author of The Price of Global Health, argues that a ‘perfect storm’ is approaching as disease burdens rise and drug budgets fall. But pharma can navigate this by thinking carefully about its value proposition, understanding the decision makers – and telling better stories.

“We are heading into a perfect storm with regard to market access and pricing issues,” warns Ed Schoonveld, Managing Principal, Market Access & Pricing at ZS. The cost of healthcare is rising rapidly while payers are under real budget pressure. “We have, for example, tremendous obesity issues globally now and because of that we have increased rates of diabetes and cardiovascular disease and we are heading into some Alzheimer’s disease treatment challenges,” he begins. “We’ve made a lot of progress, but it’s not like we have a cure for cancer either. A lot of cost increases in the system will continue to plague us.”
It is a stark comment from one of the foremost experts in this area and is sure to make pharma executives sit up and take notice. But help is at hand: Schoonveld’s book The Price of Global Health, the second edition of which has just been published, sets out frameworks which seek to show how to navigate these choppy waters. In an interview with eyeforpharma, he explained these are designed to help pharma get a cross-functional debate going.
Understanding the parameters
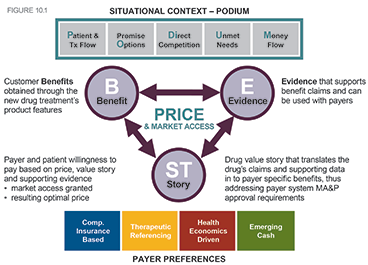
 Schoonveld’s Best Price framework (see Figure 1 below) is easy to implement, he says, giving pharma companies a systematic evaluation of payer-related needs in key areas such as what Phase III evidence is required, how to shape your drug’s value story and guidance on optimal global pricing strategy. In essence, it lays out that you must understand precisely the parameters of the therapy area you are in: what are the unmet needs, what would be a direct comparison for the value proposition you are putting forward, who is paying for what?
Schoonveld’s Best Price framework (see Figure 1 below) is easy to implement, he says, giving pharma companies a systematic evaluation of payer-related needs in key areas such as what Phase III evidence is required, how to shape your drug’s value story and guidance on optimal global pricing strategy. In essence, it lays out that you must understand precisely the parameters of the therapy area you are in: what are the unmet needs, what would be a direct comparison for the value proposition you are putting forward, who is paying for what?
In that context, pharma needs to be particularly clear about the benefit it is bringing to the table for payers, their advising clinicians – and for patients. The evidence you put forward must be able to tie in how a cancer survival benefit, say, translates into a cost-effectiveness benefit. It must also be able to demonstrate how it would fit in to Germany’s health system, or into a healthcare plan in the US. If you can make those sorts of calculations, you ought to be able to predict how successful you are likely to be in terms of market access and pricing. “It’s that benefits assessment, together with ensuring that the evidence is in place, that’s where a lot of emphasis of the discussion needs to take place,” Schoonveld adds.
Figure 1
The scale of the challenge
Globally, payers have been subject to tremendous budget challenges over the last few years. The combination of lack of funding and increasing healthcare needs from the population has been particularly acute in Europe, where a crisis in the Eurozone has made investors wary. Add that to the introduction of Obamacare in the US, AMNOG in Germany, and the debate over value-based assessment in the UK to name but three, and it is clear that the market access and pricing landscape is changing radically. On top of this, high profile public health scares - Ebola, H1N1 and anthrax among them - have all seen manufacturers and governments scrambling for solutions. “In all these cases, it doesn’t get any easier,” Schoonveld told eyeforpharma. “Meanwhile the demand for new drugs is higher than ever.”
Building your value proposition
Since payers are increasingly under pressure, there is a need to redefine the ways they spend their budget - or to accept them taking tough action in simply cutting it, which is not an attractive proposition for anyone. “Payers are going to be faced with some really tough decisions,” he goes on. “When payers are becoming more critical, you have to make sure that you have a very strong value proposition. If you don’t understand how payers are making decisions, then you are making the wrong development decisions.” The knowledge you must build up will influence such fundamental decisions as the indication you are attempting to serve and what the trial design will be. Payers rely on review committees of experts and pharma needs to make sure that it can show where a new product fits into the treatment space and why it is a better solution than what is already being used.
Beyond demonstrating value, it is critical to gain the support of the clinical community and to know how to manoeuvre the complexities of the global market. “The launch of Sovaldi may be a harbinger of change,” he goes on. Gilead Sciences’ drug has brought real innovation, virtually curing hepatitis C. “Payers are not challenging that value proposition at all,” Schoonveld adds. “What they are struggling with is the tremendous short-term increase in their drug budget.”
What not to do
We need to find ways of taking advantage of real-world data when it’s there but make sure we are not using it as a mechanism to delay access to new innovations that patients are waiting for".
For pharma, it is critical to open a direct dialogue, although Schoonveld accepts this can be hard to do directly. Experts in teams in larger companies understand payers very well – but to ensure that this expertise trickles down into firms is much harder, he says, not least because changes in what is required are taking place so rapidly. “Everything is data-driven,” he goes on, suggesting that global pricing and market access is sometimes hard to explain to departments such as R&D but that it is necessary to do so. Common mistakes by pharma include choosing the wrong comparator in a trial program or not having a direct comparator altogether. Equally, not substantiating evidence of benefit across the whole patient population can be problematic and there are other trade-offs to make.
“People should know that a placebo-controlled trial doesn’t cut it with a lot of payers,” Schoonveld adds. In oncology, clinicians are enthusiastic about positive response rates - but payers are less so. Instead, they want to see evidence of overall survival and measuring this generally requires a much bigger sample size. Progression-free survival lies somewhere in the middle, he suggests, but these are exactly the sorts of calculations pharma companies must make between getting clinician support on the one hand, and not making a trial so big that it costs too much and takes five years longer, on the other. “That’s complex, particularly in a competitive environment,” Schoonveld comments.
The importance of real-world data
Real-world data gives you a much more reliable measure of a drug’s effect on patients, and for that reason it is of great interest to regulators and payers. But it is, of course, difficult for manufacturers to present good real-world data at launch. “We need more work to be done to use real-world data as post-launch confirmation,” Schoonveld suggests. “We need to find ways of taking advantage of real-world data when it’s there but make sure we are not using it as a mechanism to delay access to new innovations that patients are waiting for.” A benefits analysis across patient groups is also critical, he says, as payers are - for some high-cost specialty drugs in particular – putting more emphasis on making sure the treatment is being used for the right patients. It is worth remembering, too, that there will be very little leeway to have your price reassessed once it has been agreed. For instance, if you set the figure at 60% of what you wanted because you don’t have the full data package at launch, it is unlikely that you will be able to raise it when the full data package becomes available. “There is no way you’re going to dramatically increase your price,” Schoonveld concludes. “You need to get it right at launch.”
Market Access & Pricing Excellence 2015
Co-create solutions with payers. Get reimbursed for real patient value.
